Removing the greenhouse gas CO2 from the atmosphere and turning it into stable carbon –a new combined facility at Karlsruhe Institute of Technology (KIT) accomplishes this on a testing scale. The NECOC research project collaborated with industry partners to develop the process that combines negative emissions with the production of a high-tech resource. As a next step, it will be optimized energetically and scaled up. The Federal Ministry for Economics and Climate Action funds the project with 1.5 million euro.
Germany is progressing on its way to climate neutrality – and has to close carbon cycles in its industries as soon as possible to get there. To reach the 1.5-degree target, the Intergovernmental Panel on Climate Change (IPCC) suggests to remove and permanently store already emitted CO2. “We have to find completely new technological solutions if we want to keep up industrial production,” says Dr. Benjamin Dietrich of the KIT Institute of Thermal Process Engineering (TVT). ”This includes the industrial carbon supply. Carbon is needed for the production of batteries, building materials, colors, and in the agricultural sector. So far, it comes largely from fossil sources.” In the research project NECOC (short for: NEgative CarbOn Dioxide to Carbon) coordinated by Dietrich, the associated partners KIT, INERATEC, and Climeworks develop a process to convert CO2 from the atmosphere into carbon. “If this carbon remains permanently bound, we successfully combine negative emission with a component of the post-fossil resource supply as part of a future carbon management strategy. This represents a double contribution to a sustainable future,” Dietrich explains. In the first project phase, the research team constructed a container-sized test facility, which now went into operation. This first-phase installation removes two kilograms of CO2 from the ambient air in one day and turns it into 0.5 kilogram of solid carbon.
In Three Steps from Greenhouse Gas to Useful Resource
The NECOC process combines three steps: The first is an absorber to separate the CO2 from the ambient air (Direct Air Capture). In the second step, the CO2 is moved to a microstructured reactor, where it reacts with sustainably produced hydrogen from a connected electrolyzer. Its components, carbon and oxygen, form new bonds and the CO2 becomes methane and water. While the water flows back to the electrolyzer, the methane including the carbon ends up in a reactor with liquid tin. This is where the third process step takes place: In rising bubbles, a pyrolysis-reaction splits the methane molecules, creating hydrogen, which can be returned to split CO2. The only remaining part is carbon, which floats on the tin as micro granular that can be taken off mechanically on a regular basis. Changing process parameters like the temperature level allows the production of different carbon modifications like graphite, carbon black, or even graphene.
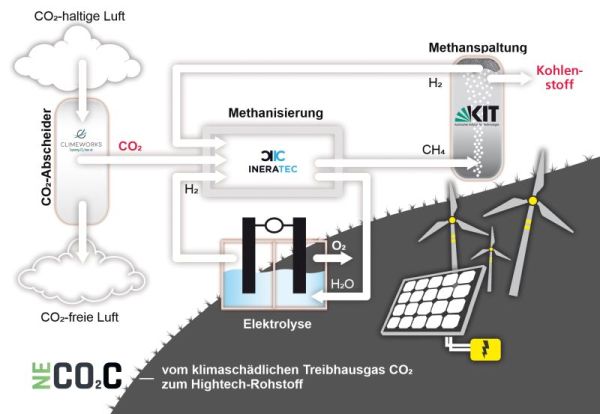
(Graph: modus: medien + kommunikation gmbh)
Optimize and Scale for Industrial Application
The start of the test installation is an important milestone for the NECOC project, as well as the end of the first funding phase. In a second project phase, the NECOC procedure will now be scaled up and optimized for expansion. “We are planning to make the procedure more energy-efficient by improving the energy recovery from the process heat,” states project director Dr. Leonid Stoppel from the Karlsruhe Liquid Metal Laboratory (KALLA). “We are also looking into an integration of high-temperature heat storages and direct solar heating.” Additional points of research are the inclusion of CO2 point sources, novel approaches to the extraction of CO2 from the air, and the influence of trace components and impurities in the process network on the carbon quality.
About NECOC
In the framework of the NECOC research project, a climate-friendly procedure with negative emissions is developed for the production of the high-tech resource carbon from atmospheric CO2 as an element of a carbon management strategy. NECOC started at the end of 2019 with the construction of the components for the three underlying process steps: Direct Air Capture, methanization, and pyrolysis. After the successful testing of each individual component, the combined installation was realized in 2022 and put into operation at the beginning of December. Involved in the project are the Karlsruhe Liquid Metal Laboratory (KALLA) as part of the Institute for Thermal Energy Technology and Safety (ITES), as well as the Institute of Thermal Process Engineering (TVT). Project partners are INERATEC GmbH, a KIT spinoff, and Climeworks Deutschland GmbH.
More Information (in German): https://www.tvt.kit.edu/21_3547.php
More about the KIT Energy Center
In close partnership with society, KIT develops solutions for urgent challenges – from climate change, energy transition and sustainable use of natural resources to artificial intelligence, sovereignty and an aging population. As The University in the Helmholtz Association, KIT unites scientific excellence from insight to application-driven research under one roof – and is thus in a unique position to drive this transformation. As a University of Excellence, KIT offers its more than 10,000 employees and 22,800 students outstanding opportunities to shape a sustainable and resilient future. KIT – Science for Impact.


