A new type of lithium-metal battery reaches an extremely high energy density of 560 watt-hours per kilogram – based on the total weight of the active materials – with a remarkable stability. Researchers of the Helmholtz Institute Ulm (HIU) of Karlsruhe Institute of Technology (KIT) used a promising combination of cathode and electrolyte: The nickel-rich cathode enables storage of high energy per mass, the ionic liquid electrolyte ensures largely stable capacity over many cycles. This is reported by the scientists in Joule (DOI: 10.1016/j.joule.2021.06.014).
Currently, lithium-ion batteries represent the most common solution for mobile power supply. In some applications, however, this technology reaches its limits. This especially holds for electric mobility, where lightweight and compact vehicles with large ranges are desired. Lithium-metal batteries may be an alternative. They are characterized by a high energy density, meaning that they store much energy per mass or volume. Still, stability is a problem, because the electrode materials react with conventional electrolyte systems.
Researchers of Karlsruhe Institute of Technology (KIT) and the Helmholtz Institute Ulm for Electrochemical Energy Storage (HIU) have now found a solution. As reported in Joule, they use a promising new combination of materials. A cobalt-poor, nickel-rich layered cathode (NCM88) reaches a high energy density. With the usually applied, commercially available organic electrolyte (LP30), however, stability leaves a lot to be desired. Storage capacity decreases with an increasing number of cycles. Professor Stefano Passerini, Director of HIU and Head of the Electrochemistry for Batteries Group, explains the reason: “In the electrolyte LP30, particles crack on the cathode. Inside these cracks, the electrolyte reacts and damages the structure. In addition, a thick mossy lithium-containing layer forms on the anode.” For this reason, the scientists used a non-volatile, poorly-flammable, dual-anion ionic liquid electrolyte (ILE) instead. “With the help of ILE, structural modifications on the nickel-rich cathode can be reduced significantly,” says Dr. Guk-Tae Kim from the Electrochemistry for Batteries Group of HIU.
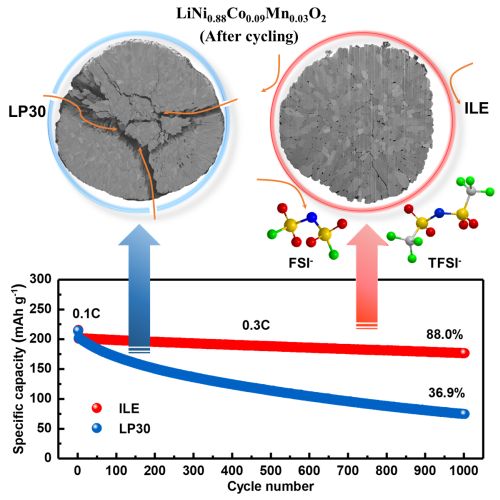
The ionic liquid electrolyte ILE (right) largely prevents structural modifications on the nickel-rich NCM88 cathode. After 1000 cycles, battery capacity still reaches 88 percent. (Figure: Fanglin Wu and Dr. Matthias Künzel, KIT/HIU)
Capacity Keeps 88 Percent after 1000 Cycles
The results: The lithium-metal battery with the NCM88 cathode and the ILE electrolyte reaches an energy density of 560 watt-hours per kilogram (Wh/kg) – based on the total weight of the active materials. Its initial storage capacity is 214 milliampere hours per gram (mAh g-1) of the cathode material. After 1000 cycles, 88 percent of the capacity are retained. The average Coulombic efficiency, i.e., the ratio between discharge and charge capacity, is 99.94 percent. As the battery is characterized by a high safety, the researchers have made an important step towards carbon-neutral mobility.
About the Helmholtz Institute Ulm
The Helmholtz Institute Ulm (HIU) was established in January 2011 by Karlsruhe Institute of Technology (KIT), Member of the Helmholtz Association, in cooperation with Ulm University. With the German Aerospace Center (DLR) and the Center for Solar Energy and Hydrogen Research Baden-Württemberg (ZSW), two other renowned institutions are involved in the HIU as associated partners. The international team of about 130 scientists at HIU works on the development of fundamentals of future energy storage systems for stationary and mobile use.
Original Publication (Open Access)
Fanglin Wu, Shan Fang, Matthias Kuenzel, Angelo Mullaliu, Jae-Kwang Kim, Xinpei Gao, Thomas Diemant, Guk-Tae Kim, and Stefano Passerini: Dual-anion ionic liquid electrolyte enables stable Ni-rich cathodes in lithium-metal batteries. Joule. Cell Press, 2021. DOI: 10.1016/j.joule.2021.06.014
https://doi.org/10.1016/j.joule.2021.06.014
More about HIU: https://hiu-batteries.de/en/

