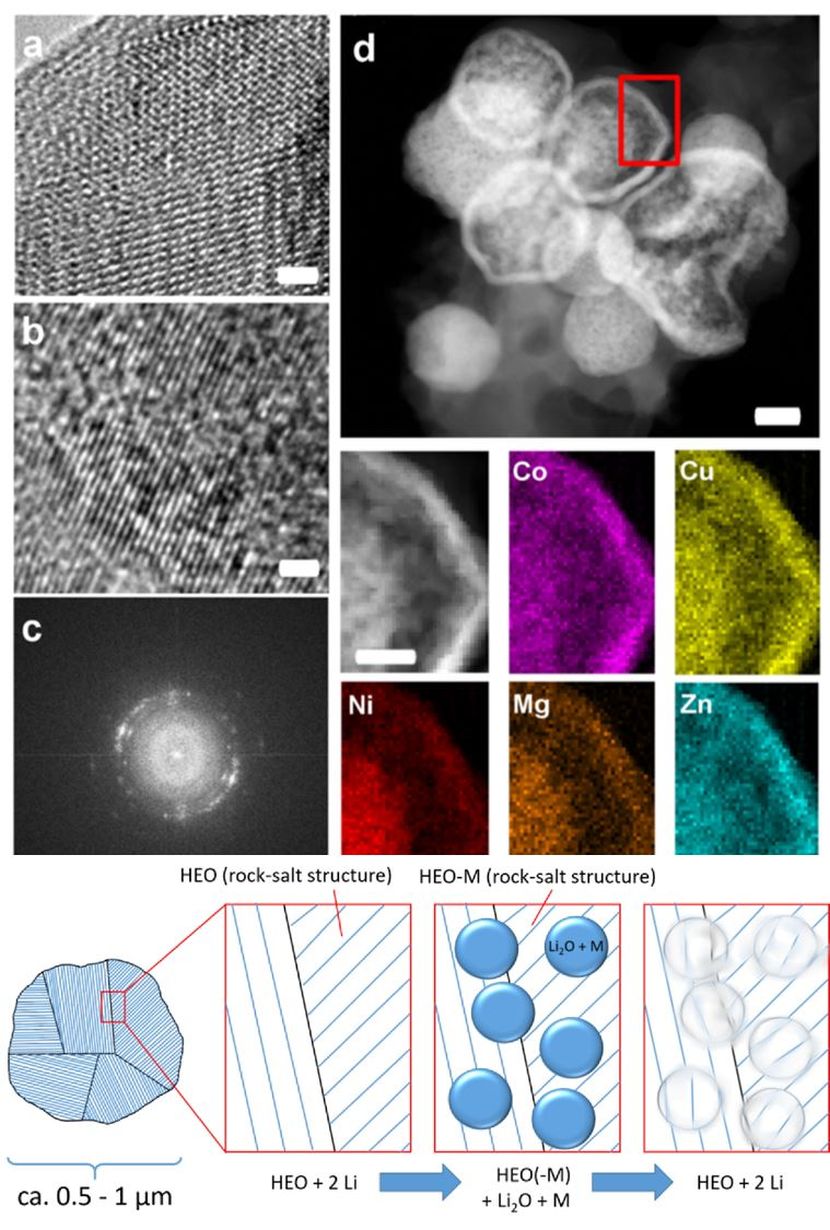
Novel materials can considerably improve storage capacity and cycling stability of rechargeable batteries. Among these materials are high-entropy oxides (HEO), whose stability results from a disordered distribution of the elements. With HEO, electrochemical properties can be tailored, as was found by scientists of the team of nanotechnology expert Horst Hahn at Karlsruhe Institute of Technology (KIT). The researchers report their findings in the journal Nature Communications. (Open Access, DOI: 10.1038/s41467-018-05774-5)
Sustainable energy supply requires reliable storage systems. Demand for rechargeable electrochemical energy storage devices for both stationary and mobile applications has increased rapidly in the past years and is expected to continue to grow in the future. Among the most important properties of batteries are their storage capacity and their cycling stability, i.e. the number of possible charging and discharging processes without any loss of capacity. Thanks to its high stability, an entirely new class of materials called high-entropy oxides (HEO) is expected to result in major improvements. Moreover, electrochemical properties of HEO can be customized by varying their compositions. For the first time, scientists of KIT’s Institute of Nanotechnology (INT) and Karlsruhe Nano Micro Facility (KNMF), of the Helmholtz Institute Ulm (HIU) established jointly by KIT and Ulm University, and of the Indian Institute of Technology in Madras have now demonstrated the suitability of HEO as conversion materials for reversible lithium storage. Conversion batteries based on electrochemical material conversion allow for an increase of the stored amount of energy, while battery weight is reduced. The scientists used HEO to produce conversion-based electrodes that survived more than 500 charging cycles without any significant degradation of capacity. They present their work in the journal Nature Communications.
The “Nanostructured Materials” group of Professor Horst Hahn, Director of KIT’s INT, is among the pioneers of research into high-entropy oxides. The scientists published several of the still rare publications about these novel materials that have been known for a few years only. The special properties of HEO result from entropy stabilization. This makes them comparable to the already better known high-entropy alloys. Entropy-stabilized HEO are complex oxides containing five or more different metal cations of the same amounts and exhibiting a single-phase crystal structure. Although typical crystal structures of the elements differ considerably, they form a joint lattice and distribute to the positions in the crystal without any apparent order. This disorder, also referred to as high entropy, stabilizes the material, probably because it impairs migration of defects in the lattice.
“Thanks to the high stability, the interactions of the different metal cations, and the high number of feasible element combinations, HEO open up undreamt-of opportunities,” says Professor Horst Hahn. The study presented in Nature Communications concentrated on HEO based on transition metals (TM-HEO), which are characterized by high lithium ion conductivity. By means of transmission electron microscopy (TEM), the researchers studied the structure of TM-HEO and its impact on the conversion reaction. They found that removal of one element only reduces entropy and adversely affects cycling stability. Each individual element influences the electrochemical behavior of the TM-HEO, such that the materials can be tailored to various applications. The result is a modular approach to the systematic development of electrode materials. “Our study has shown that entropy-stabilized HEO considerably differ from classical conversion material,” Horst Hahn says. “To open up their full potential for energy storage applications, however, further research is needed.”
Originalpublication (Open Access):
Abhishek Sarkar, Leonardo Velasco, Di Wang, Qingsong Wang, Gopichand Talasila, Lea de Biasi, Christian Kübel, Torsten Brezesinski, Subramshu S. Bhattacharya, Horst Hahn & Ben Breitung: High entropy oxides for reversible energy storage. Nature Communications. 2018. DOI: 10.1038/s41467-018-05774-5
More about the KIT Energy Center: http://www.energy.kit.edu
Being “The University in the Helmholtz Association”, KIT creates and imparts knowledge for the society and the environment. It is the objective to make significant contributions to the global challenges in the fields of energy, mobility, and information. For this, about 10,000 employees cooperate in a broad range of disciplines in natural sciences, engineering sciences, economics, and the humanities and social sciences. KIT prepares its 22,800 students for responsible tasks in society, industry, and science by offering research-based study programs. Innovation efforts at KIT build a bridge between important scientific findings and their application for the benefit of society, economic prosperity, and the preservation of our natural basis of life. KIT is one of the German universities of excellence.