Chemical reactions can be initiated through exposure to light. An international research group led by KIT uses this effect to switch on or off chemical reactions using light. This allows different target products to be produced alternately in one single reaction vessel from the same source material – simply by switching light on and off. They have now presented their experiments with polymers in the Journal of the American Chemical Society. (DOI: 10.1021/jacs.6b01805)
“The key to the method lies in its simplicity,” says Professor Christopher Barner-Kowollik from the Institute for Chemical Technology and Polymer Chemistry (ITCP) at KIT. “Traditionally, chemical reactions can be triggered through heat or light. Now we can choose and control which bonds the reacting substances form,” the chemist, who headed up the international research group together with Dr. James Blinco from the Queensland University of Technology, explains.
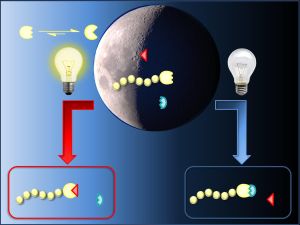
The concept is simple. Substance A reacts with substance B when light is excluded, but with substance C when exposed to light. If you put all three source materials in one single reaction vessel, the reaction can be controlled through the presence or absence of light depending on the desired target product. “We're demonstrating that the chemical reaction can easily be manipulated in both directions through light,” says Barner-Kowollik. “The choice of bonding partner is only determined by the external light field without the need to add other reagents,” Barner-Kowollik points out. The chemist is head of the Division of Preparative Macromolecular Chemistry at KIT and deals with the synthesis of macromolecular chemical bonds.
The process, which has now been presented for the first time in the prestigious US Journal of the American Chemical Society under the title “A Light-Activated Reaction Manifold”, was examined using the formation of small molecules and polymers as an example. A variety of products could easily be obtained within a single production process in industrial applications thanks to the new method involving a light-controlled sequence of reactions. Another option would be, according to Barner-Kowollik, to assign different properties to a functional surface during its production by having only certain areas react to the exposure to light.
Along with the members of ITCP and the Institute for Biological Interfaces at KIT, scientists from Ghent University, Belgium, and Queensland University of Technology (QUT), Australia, where Barner-Kowollik also holds a chair, are involved in the research. “Transnational research requires a high level of coordination, but you can achieve a lot if you combine know-how across the world,” says the KIT scientist. This research into light-controlled chemical reactions was funded by the Chemical Industry Fund, the Helmholtz Association, and the QUT, among others.
For more information go to:
http://www.chemistryviews.org/details/ezine/9380101/The_Light_and_Dark_Sides_of_Chemistry.html
Kai Hiltebrandt, Katharina Elies, Dagmar R. D’hooge, James P. Blinco, and Christopher Barner-Kowollik: A Light-Activated Reaction Manifold. Journal of the American Chemical Society (2016). DOI: 10.1002/chemv.201600047, http://pubs.acs.org/doi/abs/10.1021/jacs.6b01805
Being “The University in the Helmholtz Association”, KIT creates and imparts knowledge for the society and the environment. It is the objective to make significant contributions to the global challenges in the fields of energy, mobility, and information. For this, about 10,000 employees cooperate in a broad range of disciplines in natural sciences, engineering sciences, economics, and the humanities and social sciences. KIT prepares its 22,800 students for responsible tasks in society, industry, and science by offering research-based study programs. Innovation efforts at KIT build a bridge between important scientific findings and their application for the benefit of society, economic prosperity, and the preservation of our natural basis of life. KIT is one of the German universities of excellence.